Iodoform Test for Alcohols
Highly resistant to most acids alcohols detergents and solvents. Give the reactions chemical equation of test samples with iodoform.
If Acetone And Alcohol Both Give A Positive Result In An Iodoform Test How Do We Identify Whether It Is Given By Alcohol Or Acetone Quora
Organic Compounds with Functional Groups Containing Oxygen and Nitrogen.

. A pale yellow crystalline volatile substance it has a penetrating and distinctive odor in older chemistry texts the smell is sometimes referred to as that of hospitals where the compound is still commonly used and. Iodoform test for alcohols containing the. A spectacular practical demonstration to show the differences between elements.
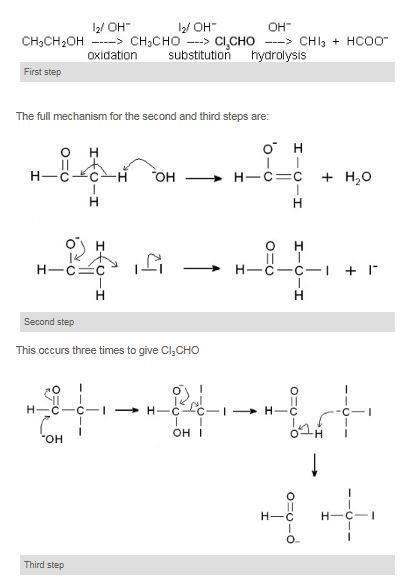
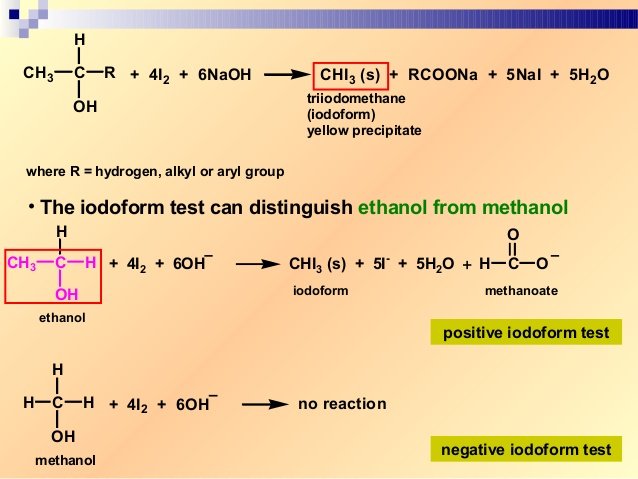
A question about qualitative analysis which is to be accomplished. O-nitrophenol is more volatile. The reaction of iodine a base and a methyl ketone gives a yellow precipitate along with an antiseptic smellIt also tests positive for a few specific secondary alcohols that contain at least one methyl group.
View all the details of MET 2022 Exam such as exam dates registration admit card syllabus eligibility result question papers cut off counselling etc. Check the chemical compatibility of LDPE low density polyethylene with various chemicals solvents alcohols and other products. When fumarate reacts with D2O in the presence of the enzyme fumarase only one isomer of the product.
Alcohols Phenols and Ethers. Chemical Compatibility Chemical Compatibility Alkazene D-Severe Effect AlcoholsEthyl B-Good Ammonia Gas Cold A-Excellent Alcohols. Take 1ml of given organic compound in a clean dry test tube.
Furfuryl D-Severe Effect Ammonia Gas Hot A-Excellent AlcoholsHexyl B-Good. A test to distinguish between ethanol and methanol. Here the alkyl halide should be primary and alkoxide should be tertiary.
Students who are preparing for their Class 12 exams must go through NCERT Solutions for Class 12 Chemistry Chapter 10 Haloalkanes and HaloarenesGoing through the solutions provided on this page will. Add dilute sodium hydroxide solution drop wise until the brown colour of iodine is discharged. The CUETCUCET syllabus 2022 is the same as the Class 12 syllabus.
Nomenclature methods of preparation physical and chemical properties of primary. Iodoform also known as triiodomethane and inaccurately as carbon triiodide is the organoiodine compound with the chemical formula C H I 3. The reaction between zinc powder and sulfur.
Haloalkanes And Haloarenes Class 12 NCERT Solutions includes all the important topics with detailed explanation that aims to help students to understand the concepts better. NEET Online Test Series. Alcohols Phenols and Ethers.
Before permanent installation test the equipment with the chemicals and under the specific conditions of your application. Ideal for use with reactive and corrosive chemicals. Propan-2-ol gives yellow ppt of iodoform whereas methanol does not.
LDPE is defined by a density range of 09100940 gcm3. Electronic structure important methods of preparation important reactions and physical properties of alcohols phenols ethers aldehydes ketones carboxylic acids nitro compounds amines diazonium salts cyanides and isocyanides Specific. Which of the following isomers is more volatile.
The melting point of PTFE 1 is 600K 327C 620F Low temperature. Maintains high strength toughness and self-lubrication as low as 5K -268C -450F and good flexibility at 194K -79C -110F. This test is given by secondary alcohols ketones and acetaldehyde.
Or collect it over water and test it with a lighted spill. -glucose -fructose Give the A. Ceric ammonium nitrate test.
JEE Mains Chapter wise Practice Questions Last 30 Years with 5000 Questions for online practice. A class practical to distinguish between methanol and ethanol using the iodoform reaction. Add 1ml of 1 iodine solution to it.
Sodium metal test This test is based on the appearance of effervescence due to liberation of hydrogen gas when the alcohol is reacted with active metals like sodium. Includes kit list safety instructions procedure and teaching notes. Ethanol when reacted with I2 and NaOH or NaOI gives yellow ppt of iodoform since it has the presence of CH3-CH OH- group.
Organic Chemistry- Some Basic Principles and Techniques. B From alkyl halide and sodium alkoxide. 2014 Very Short Answer Type Questions 1 Mark Question 21.
Hence students who have prepared and appeared for the Board exams or the other national-level competitive entrance exams like JEE Main or NEET would find it easy to. Iodoform test is used to check the presence of carbonyl compounds with the structure R-CO-CH 3 or alcohols with the structure R-CHOH-CH 3 in a given unknown substance. The information in this chart has been supplied to Cole-Parmer by other reputable sources and is to be used ONLY as a guide in selecting equipment for appropriate chemical compatibility.
Request a small 3 x 3 silicone membrane test sample within the US only. Candidates can go through the same to get a brief idea of the topics that will be covered in the Common Entrance Test. It is not reactive at room temperatures except by strong oxidizing agents and some solvents cause swelling.
Effect of substituents on alpha- carbon on. NEET Past Year Papers. To illustrate the differences in reactivity of isomeric alcohols try using the iodoform demonstration in the video.
Get in touch with us. Short Answer Type Questions I 2 Marks Question 23. In association with Nuffield Foundation.
LDPE Chemical Compatibility Chart.
A Test To Distinguish Between Ethanol And Methanol Experiment Rsc Education

Alcohols Advanced 9 Iodoform Test For Ch3ch Oh R Youtube

Iodoform Test Description And Mechanism Compounds That Test Positive

Iit Jee Iodoform Test For Alcohols Offered By Unacademy

Iodoform Test Description And Mechanism Compounds That Test Positive
Comments
Post a Comment